Tuesday, October 22, 2013
Vitamin D Levels Not Linked to Kidney Stone
ACP issues clinical practice guideline on chronic kidney disease
ACP released a new clinical practice guideline this week on screening, monitoring and treatment of stage 1 to 3 chronic kidney disease (CKD).

The guideline was based on a systematic evidence review that evaluated the relevant English-language literature published from 1985 through November 2011. The clinical outcomes evaluated were all-cause mortality, cardiovascular mortality, composite renal outcomes, end-stage renal disease, quality of life, physical function and activities of daily living. The guideline was published online byAnnals of Internal Medicine on Oct. 22.
ACP's recommendations are as follows:
- Recommendation 1: ACP recommends against screening for CKD in asymptomatic adults without risk factors for CKD. (Grade: weak recommendation, low-quality evidence)
- Recommendation 2: ACP recommends against testing for proteinuria in adults with or without diabetes who are currently taking an ACE inhibitor or an angiotensin II receptor blocker. (Grade: weak recommendation, low-quality evidence)
- Recommendation 3: ACP recommends that clinicians select pharmacologic therapy that includes either an ACE inhibitor (moderate-quality evidence) or angiotensin II receptor blocker (high-quality evidence) in patients with hypertension and stage 1 to 3 CKD. (Grade: strong recommendation)
- Recommendation 4: ACP recommends that clinicians choose statin therapy to manage elevated low-density lipoprotein in patients with stage 1 to 3 CKD. (Grade: strong recommendation, moderate-quality evidence)
The guideline authors also gave advice to clinicians on provision of high-value care in this population, noting that no evidence shows that screening improves clinical outcomes in adults without risk factors and that there is no proven benefit of screening in adults already taking ACE inhibitors or angiotensin II receptor blockers for microalbuminuria.
"In the absence of evidence that screening improves clinical outcomes, testing will add costs, owing to both the screening test and to additional follow-up tests (including those resulting from false-positive findings), increased medical visits, and costs of keeping or maintaining health insurance," the guideline authors concluded.
Thursday, October 17, 2013
Flaxseed May Reduce Blood Pressure in Hypertensive Patients
Poor Glycemic Control a Major Factor in Overestimating GFR in Diabetics
BMI-Mortality Link Significant, but Small
Action Points
- Body mass index was the least predictive risk factor for mortality over 10 years in obese patients who were eligible for bariatric surgery, a study found.
- Note that obese patients were more likely to be at risk for death for each year of age, if they were male, if they were smokers, or if they had diabetes.
Among predictors of mortality in obese patients, BMI was significantly predictive, but just barely (OR 1.03, 95% CI 1.01-1.05), according to Raj Padwal, MD, MSc, of the University of Alberta in Edmonton, and colleagues.
Obese patients were more likely to be at risk for death for each year of age (OR 1.09 per year, 95% CI 1.07-1.10), if they were male (OR 1.50, 95% CI 1.20-1.87), if they were smokers (OR 1.62, 95% CI 1.28-2.06), or if they had diabetes (OR 2.25, 95% CI 1.76-2.87), they wrote online in the Oct. 16 issue of JAMA Surgery.
The study adds to an already vast body of research on the effects of BMI on morbidity and mortality.
In past research, outcomes have been mixed where some studies haveassociated risks of death with BMI, especially low BMI, while other research has shown that a higher BMI was protective for hearts in some populations.
Other research has shown that a pregnant mother's obesity was tied to health problems and risks for early mortality for her child, while another study found that there were no extra risks of death tied to moderate obesity in particular.
However, distribution of weight, such as at the belly, has been tied to increased risks for cancer, heart disease, and death.
In the current study, the authors looked at mortality predictors among 15,394 obese English patients whose BMI made them eligible for bariatric surgery.
All participants were registered to the English General Practice Research Database, a database of anatomized, linked, longitudinal medical records in a population-representative sample of 6% of the English population. The records include patient demographics, health behaviors, physiological and laboratory data, clinician-assigned diagnoses, outpatient prescription medications, and disease registries for hypertension and diabetes.
Patients were ages 18 to 65 and had a BMI of 35 or greater, or a BMI of 30 to 34.9 and an obesity-related comorbidity, which included hypertension, dyslipidemia, heart failure, diabetes, sleep apnea, osteoarthritis, coronary artery disease, and cerebrovascular disease.
Cohort entry date was considered the first time a patient's record indicated that their BMI made them surgery-eligible.
The primary outcome was 10-year all-cause mortality in relation specifically to age, sex, BMI, smoking and alcohol intake, and chronic medical conditions.
Over the 10-year period, 2.1% of participants died, and "within-age strata did not differ significantly between BMI classes."
Among comorbidities, age, diabetes, male sex, smoking status, coronary artery disease, cerebrovascular disease, and BMI were predictive of 10-year mortality. Age, diabetes, smoking status, and male sex were the comorbidities with a P=0.99 overall goodness fit.
The authors noted in their conclusion that, although they were significant, "established cerebrovascular disease and BMI did not contribute materially to the mortality prediction in the primary analysis."
They added that the study was limited by lack of analysis of other clinically important end points separate from all-cause mortality, potential under-reporting of comorbidity prevalence, results that may not generalize outside of England, and use of BMI as recorded at a single point in time.
The study was supported by the Canadian Institutes of Health Research.
The authors received support from the Government of Alberta, the University of Alberta, Alberta Health Services Chair in Obesity Research and Management, the Alberta Heritage Foundation for Medical Research, Alberta Innovates-Health Solutions, Ethicon Endo-Surgery, Coviden, Gore, Johnson and Johnson, Bard, Baxter, and Olympus.
Primary source: JAMA Surgery
Source reference: Padwal RS, et al "A simple prediction rule for all-cause mortality in a cohort eligible for bariatric surgery" JAMA Surg 2013; DOI: 10.1001/jamasurg.2013.3953.
Wednesday, October 16, 2013
Antihypertensives Help Prevent Cardiovascular Events in CKD Patients
Diabetics Should Receive Same Diet and Lifestyle Advice as Others
Saturday, October 12, 2013
Fish Oil for Prevention of Sudden Death in Hemodialysis Patients?
Fish Oil for Prevention of Sudden Death in Hemodialysis Patients?
Daniel Teta
DisclosuresKidney Int. 2013;83(6):993-995.
Friedman et al. report that hemodialysis patients with the highest levels of n-3 fatty acids had impressively low odds of sudden cardiac death. The study is limited by a small sample size, and the analysis relies on only a single baseline measurement of blood levels. Recent randomized evidence indeed fails to support that n-3 fatty acids may prevent sudden death in nonrenal patients. More evidence is needed to advocate fish oil in this setting.
Dialysis patients have dramatically low survival rates, between 35 and 45% at 5 years. Cardiovascular disease is responsible for 43% of deaths in this setting, and sudden cardiac death has now emerged as the leading single cause of mortality, accounting for approximately 25% of deaths. Sudden death is defined as an unexpected 'natural' death within a very short time period in a person without any prior condition that would appear fatal. Data from the general population indicate that about 80% of sudden deaths may be due to ventricular fibrillation. The mechanisms causing the extraordinarily excessive rate of sudden deaths in dialysis patients are largely unknown. A variety of kidney- and dialysis-specific factors may favor the occurrence of this complication, including left ventricular hypertrophy, heart failure, volume overload, myocardial fibrosis, hyperkalemia, hyperphosphatemia, QT dispersion, QT-prolonging medication, sympathetic overactivity/autonomic nerve dysfunction, and electrolyte and volume shifts during hemodialysis (HD) sessions.[1] Because sudden death may derive from many causes, its prevention is a difficult challenge for nephrologists. Successful preventive strategies applied in the general population, such as the use of β-blockers, angiotensin-converting enzyme inhibitors, angiotensin II receptor antagonists, statins, and implantable cardioverter-defibrillators, do not appear to offer benefit in dialysis patients.[1]In this pessimistic context, novel routes targeting the risk of sudden death in this population are most welcome.
Long-chain n-3 fatty acids, also called ω-3 fatty acids, are polyunsaturated fatty acids commonly found in marine and plant oils. Their chemical structure includes a double bond (C=C), starting after the third carbon atom from the end of the carbon chain. These fatty acids are essential nutrients for cell-membrane structure and physiological functions such as platelet aggregation and lipid metabolism. A major role for n-3 fatty acids was suggested in the 1970s when extremely low rates of death from cardiovascular disease were reported among the Inuit, as compared with northern European counterparts, despite similar high intakes of fat, about 40% of calories. High amounts of n-3 fatty acids from fish and fish-eating mammals were actually the main sources of fat among the Inuit. In the past decade, in vitro studies, animal experiments, observational studies, and some randomized controlled trials (RCTs) have demonstrated that n-3 fatty acids had beneficial effects on major cardiovascular outcomes, including sudden death, due to antiatherogenic, anti-inflammatory, antithrombotic, antihypertensive, and triglyceride-lowering effects.[2] ω-3 fatty acids have distinct antiarrhythmic effects on myocyte electrophysiology—for example, alteration of the function of membrane sodium channels, L-type calcium channels, and sodium–calcium exchangers.[2] There are three major n-3 fatty acids: eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), found in fatty fish (salmon, bluefish, mackerel, arctic char, sardines, and swordfish), and α-linolenic acid (ALA), found in specific vegetable oils (soybean, rapeseed, and flaxseed), walnuts, and some green vegetables (brussels sprouts, kale, spinach, and salad greens). The human organism may partially convert ALA to EPA and DHA, thus allowing vegans to get sufficient amounts of n-3 fatty acids. Western diets typically contain high amounts of saturated fats and insufficient amounts of n-3 fatty acids. Thus, some but not all current guidelines from major societies recommend the use of n-3 fatty acids, either through diets or as supplements.
The question of whether dialysis patients should be supplemented with n-3 fatty acids is appealing. Friedman et al. [3] (this issue) analyzed data from 100 patients who died from sudden cardiac death during their first year of dialysis treatment and 300 frequency-matched control survivors from a US cohort of HD units. Blood levels of long-chain n-3 fatty acids, measured at the time of dialysis initiation, were inversely associated with the risk of sudden cardiac death during the first year of HD, even after adjustment for multiple potential confounders. The odds of sudden cardiac death at 1 year of patients in the two highest quartiles for blood levels of long-chain n-3 fatty acids were remarkably low: 0.22 and 0.20 for the third and fourth upper quartiles, respectively, compared with the lowest quartile. Although these data are encouraging, the study has limitations, including the small size of the patient sample and the fact that the analyses were based on a single baseline measurement that may not reflect levels of n-3 fatty acids over a long period. The initiation of a dialysis program indeed determines a number of changes that may affect blood levels and profiles of n-3 fatty acids, from the baseline value. These modifications include a spontaneous increase in food intake, changes in pharmacological treatment that may interact with blood lipids, and improvement in physical activity. Interestingly, rates of hyperkalemia during the 12 months of follow-up were similar between cases of sudden death and controls. However, other variables that may have contributed to sudden deaths were not measured or recorded during the follow-up—for instance, interdialytic weight gains, electrolyte and volume shifts during HD, and smoking habits. In spite of these limitations, the study demonstrates a strong association between blood levels of n-3 fatty acids and sudden cardiac death in this population. Furthermore, the dramatically low odds of sudden cardiac death in patients from the top quartiles of blood levels of n-3 fatty acids, in the context of biological plausibility, are suggestive of causation.
The story of long-chain n-3 fatty acids is not new to nephrologists. In 1994, Donadio et al.showed that supplementation with fish oil remarkably slowed the progression of IgA nephropathy.[4] After a long period of silence in the field of nephrology, fish oil received renewed attention in 2012, thanks to a Canadian RCT in which fish oil ingestion was found to reduce thrombosis from new HD grafts and to improve cardiovascular-event-free survival in HD patients.[5] It is noteworthy that these end points were secondary outcomes of the study. The primary outcome, the proportion of grafts with loss of native patency, was not statistically improved. Taken together, these recently published studies in HD patients raised new hopes and, according to their authors, should prompt RCTs with fish oil supplementation to improve cardiovascular outcomes in this setting.
A decade ago, enthusiasm for n-3 fatty acids was sky high, with medical evidence from studies in nonrenal patients showing that ω-3s provided by food or supplements had a strong cardiovascular protective effect across all major cardiovascular outcomes.[6]Unfortunately, since then, the picture has clouded. Although epidemiological studies have consistently shown that diets rich in n-3 fatty acids or blood levels of n-3 fatty acids were associated with impressively low odds of sudden death,[7] results from prospective trials in which n-3 fatty acids were supplemented through diets or pills have provided conflicting results. The protective effect shown initially became non-significant, as more randomized evidence accumulated.[8] In 2012, a large meta-analysis including 20 RCTs and almost 70,000 patients failed to show any benefit in the prevention of major cardiovascular end points such as cardiac death, myocardial infarction, and stroke.[9] In the latter meta-analysis, sudden death was examined in seven RCTs, including 41,751 patients and 1030 events, and yet n-3 fatty acids failed to reduce the odds of sudden death, thus rejecting a distinct antiarrhythmic effect. Finally, a double-blind RCT involving 12,536 participants at high risk for cardiovascular events (patients with diabetes, impaired fasting glucose, and impaired glucose tolerance) was equally negative regarding cardiovascular events, including deaths from arrhythmias.[10] The failure to replicate earlier positive findings with n-3 fatty acids may be explained by the analysis of much larger studies, the improvement in the quality of the studies, and the systematic use of state-of-the-art concomitant treatments—antihypertensive, antithrombotic, and statins—to manage cardiovascular disease. Discrepancies between positive epidemiological evidence in favor of fish or ω-3 eaters and the absence of effect in prospective RCTs are inevitably linked to the fact that people eating a lot of fish are more likely to benefit from a healthier diet with less saturated fats from hamburgers and processed foods, than are those consuming little or no fish.
Of course, this compelling negative evidence in nonrenal patients does not exclude the possibility that n-3 fatty acids might be efficient in reducing rates of sudden death in extremely high-risk individuals, such as those initiating dialysis. Dialysis patients may indeed benefit from n-3 fatty acids in various indications (Figure 1). However, the lessons learned from studies in non-dialysis settings, coupled with the consistent history of negative trials in the dialysis population, should invite caution. Further steps may be required before investment of resources in an RCT with n-3 fatty acids in this population. First, additional epidemiological evidence linking sudden death and levels of n-3 fatty acids should be obtained from larger samples of HD patients. Second, relationships between diets and blood levels of n-3 fatty acids, and type of n-3 fatty acids, in HD patients in diverse countries and ethnic groups should be obtained, in order to target people who are more likely to benefit from supplementation. Third, a dose–response curve should be sought, in order for an interventional trial to reach blood levels of n-3 fatty acids matching those from the top quartiles in the study by Friedman et al. [3] Fourth, the safety of n-3 fatty acids should be more precisely addressed if large samples of HD patients are being tested. Although fish oil is generally harmless, it theoretically might increase the risk of bleeding especially in patients taking aspirin, clopidogrel, and anticoagulants, which are prevalent in this population. A last philosophical consideration is that miracle ω-3 pills may be no substitute for a healthier lifestyle including eating fish at least twice a week, getting lots of physical activity, and not smoking.
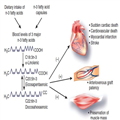
Figure 1.
Potential effects of long-chain n-3 fatty acids on outcomes in hemodialysis patients.The long-chain n-3 fatty acids eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) may be provided from dietary fish oil, and α-linolenic acid (ALA) from specific vegetable oils and some green vegetables. n-3 fatty acids can be readily provided by pharmacological supplementation as well. Both routes increase blood levels of n-3 fatty acids and may potentially lead to relevant clinical effects in hemodialysis patients, such as reduction in cardiovascular complications including sudden cardiac death, improvement in graft patency, and preservation of muscle mass. ALA may be partially converted in the organism into EPA and DHA. The plus signs indicate stimulation, and the minus signs indicate inhibition.
Colloid and Crystalloid Use for Fluid Resuscitation Leads to Similar 28-day Mortality in Patients with Hypovolemia
FDA approves medication for pulmonary hypertension.
Bloomberg News (10/9, Edney, 1.91M) reports that the Food and Drug Administration announced that it has granted approval to Bayer AG’s pulmonary hypertension drug riociguat (Adempas) for the treatment of two forms of the condition. The medication “is the first to treat the form of the condition that lasts six months or longer often caused by...chronic thromboembolic pulmonary hypertension.” The FDA said the drug is from “a class of drugs called soluble guanylate cyclase stimulators that help arteries relax to increase blood flow and decrease blood pressure.”
Reuters (10/9, Clarke) adds that the drug will include a boxed warning advising against its use in pregnant women because of the potential for fetal harm.
Forbes (10/8, 6.03M) contributor Larry Husten writes that Director of the Division of Cardiovascular and Renal Drug Products in the FDA’s Center for Drug Evaluation and Research, Norman Stockbridge, said in a press release, “Adempas is the first in its drug class approved to treat pulmonary hypertension and the first drug of any class to be shown to be effective for patients with CTEPH.” Medscape (10/9, Wood, 164K) also reports on the story.
Too Little, Too Much Sleep Linked with Increased Risk of Chronic Diseases
Hip Fracture Rates Down Among Hemodialysis Patients
10 Fall Exercise Do's and Don'ts
Why wait until January to start on a health kick or launch a new fitness routine? This season, the weather’s more moderate, so seize the day and protect your kidneys with10 simple tips!
Study Shows Benefits of Peritoneal Dialysis Catheter Placement with Omentum Management
Vitamin D Insufficiency Common and Linked with Kidney Function in CKD Patients
ICU patients may often leave with substantial cognitive deficits.
The CBS Evening News reported that, according to a study published Oct. 3 in the New England Journal of Medicine, “patients who are treated in intensive care units often leave with dementia, even when there is no evidence of it prior to their treatment.”
The Los Angeles Times (10/3, Healy, 3.07M) reports that the study revealed that almost two-thirds of ICU patients “come away from the experience with substantial mental deficits,” and that “three months after leaving the hospital, four in 10 patients continue to have cognitive problems on a par with those seen in cases of moderate traumatic brain injury. “ Approximately a quarter of ICU patients “experience a decline in mental function akin to that seen in patients with mild Alzheimer’s,” the study found.
The Tennessean (10/3, Wilemon, 417K) reports, “The longer a patient suffers from delirium while in the hospital, the higher the risk for dementia once discharged, the study” also found. What’s more, “the practice of putting critically ill patients into drug-induced comas can lengthen delirium.” Interestingly, “age was not a significant factor for the loss in mental abilities, according to the study, which also found similar results in patients” under the age of 50.
HealthDay (10/3, Thompson, 5K) points out that the study “involved 821 ICU patients” whose progress and “ability to plan and solve problems” was monitored “at regular intervals.” The story was also covered by the CBS News (10/3, Jaslow, 3.87M) website and the Wall Street Journal (10/2, A6, Landro, Subscription Publication, 5.91M).
Hospital-acquired anemia associated with increased mortality, study finds
Hospital-acquired anemia (HAA) is common and appears to be associated with increased mortality rates, according to a new study.
Researchers analyzed hospitalizations at 1 academic medical center and 9 community hospitals in the Cleveland Clinic Health System from Jan. 1, 2009, to Aug. 31, 2011, to evaluate the prevalence of HAA and to determine whether an association existed among HAA and increased mortality, length of stay (LOS) or total hospital charges. Mild HAA was defined as hemoglobin levels >11 g/dL and <12 g/dL in women and >11 g/dL and <13 g/dL in men, moderate HAA was defined as a hemoglobin level of 9.1 to ≤11.0 g/dL, and severe HAA was defined as a hemoglobin level ≤9.0 g/dL. Information on demographics, comorbid conditions and outcomes was obtained from administrative data, while data on hemoglobin values were obtained from the electronic medical record. The study appeared in the September Journal of Hospital Medicine.
Among the 188,447 hospitalizations included in the analysis, 139,807 patients (74%) developed HAA. HAA was mild in 29% of cases, moderate in 41% and severe in 30%. Patients were more likely to develop HAA if they were older and had more comorbid conditions. Risk-adjusted odds ratios for in-hospital mortality in patients with HAA were 1.0 for mild HAA (P=0.8), 1.51 for moderate HAA (P<0.001) and 3.28 for severe HAA (P<0.001). Patients with HAA also had higher risk-adjusted relative mean LOS and hospital charges compared with patients who didn't have HAA; risk-adjusted odds ratios were 1.08, 1.28 and 1.8 for LOS and 1.06, 1.18 and 1.80 for hospital charges with mild, moderate and severe HAA, respectively (P<0.001 for all comparisons).
The authors noted that their study used administrative data, involved only 1 health system and did not address treatment, among other limitations. However, they concluded, HAA is common during hospitalization and can have important effects on health care.
"Hospitals must continue to focus on improving patient safety and raising awareness of HAA and other modifiable hospital-acquired conditions," they wrote. "Closer prospective investigation for both medical and surgical patients of cumulative blood loss from laboratory testing, procedural blood loss, and a risk-benefit analysis of treatment options is necessary."
Everolimus Slows Disease Progression in Some Cases of Advanced Papillary Kidney Cancer
Leisure Time Physical Activity Linked with Reduced Risk of Hypertension
Study: Hormone Therapy may be good short-term option to relieve menopause symptoms.
The Wall Street Journal (10/2, Beck, Subscription Publication, 5.91M) reports that research (10/2, 730K) published in the Journal of the American Medical Association suggests that while hormone therapy may be a good short-term option to relieve symptoms of menopause, it should not be used to protect against chronic diseases.
USA Today (10/1, Painter, 5.82M) reports that researchers looked at data from the Women’s Health Initiative. The new study’s findings, which were “based on 13 years of follow-up and published in the Journal of the American Medical Association, show a complex mix of risks and benefits for women who take estrogen either alone or with progestin.” The researchers found that “on balance, the risks – including increases in heart disease and breast cancer for many women – outweigh the benefits when the hormones are used for anything other than relief of moderate to severe menopause symptoms.”
The New York Times (10/2, Grady, Subscription Publication, 9.61M) reports that the researchers found that “for combined hormones, for every 10,000 women taking the drugs...there were six additional instances of heart problems, nine more strokes, nine more blood clots in the lungs and nine more cases of breast cancer.” However, “there were six fewer cases of colorectal cancer, one fewer case of uterine cancer, six fewer hip fractures and one fewer death.” The majority “of the effects wore off once the drugs were stopped, but the risk of breast cancer remained slightly elevated.”
Forbes (10/1, 6.03M) contributor Larry Husten writes that among women who only received estrogen, HRT “was associated with an increased risk for stroke and venous thrombosis, and a lowered risk for hip and total fractures.” The researchers “were surprised by and at a loss to explain a small nonsignificant drop in breast cancer.” Meanwhile, “women under the age of 60 in this group also had small but signifcant reductions in all-cause mortality and myocardial infarction.”
The Boston Globe (10/2, Kotz, 1.75M) “Daily Dose” blog reports that in an accompanying editorial (10/2, 730K), Dr. Elizabeth Nabel, former director of the National Heart Lung and Blood Institute, wrote, “These findings demonstrate that menopausal hormone therapy has a complex profile of risks and benefits.” Dr. Nabel added, “Even though short-term use of hormone therapy may be useful for menopausal symptom relief ... long-term use of hormone therapy for chronic disease prevention is not warranted.” Also covering the story are Reuters (10/2, Seaman, Brooks), CNN (10/1, 14.53M), MedPage Today (10/2, Fiore, 122K), HealthDay (10/2, Norton, 5K),Medscape (10/2, Hand, 164K), and the Cleveland Plain Dealer (10/2, Townsend, 966K).
FDA approves Kcentra for the urgent reversal of anticoagulation in adults with major bleeding
Dispose of Prescription Drugs
If you have unwanted prescription drugs or over-the-counter medicines, this is a great opportunity to safely discard them.
Enter your ZIP code to find a collection site near you.
Properly disposing of medicines is important to human health and environmental protection.
- Don't flush medicines down the toilet or drain. Doing so could affect drinking water sources.
- Don't throw medicines directly in the trash. Doing so could lead to the poisoning of a child or pet, or drug abuse by a teen or adult.
- Do find out how to properly dispose of medicines (PDF).
Friday, October 11, 2013
Study: 2010 pertussis outbreak tied to vaccine refusals.
A number of national media outlets, including a network news program, report on the connection between recent pertussis outbreaks and parental vaccine refusals. On its Monday evening broadcast, the CBS Evening Newsreported on the results of a new study published in Pediatrics examined the 2010 California pertussis outbreak that sickened over 9,000 and killed ten, “found it was worst among 39 communities with clusters of kids who were not vaccinated.” According to the report, in communities with higher rates of vaccine refusal, pertussis outbreaks were twice as likely to occur. In 2010, “the percentage of parents choosing to opt out of vaccinating their children for school has tripled from .7% in 2000 to 2.3%.”
USA Today (9/30, Healy, 5.82M) reports the study used “data from the California Department of Public Health,” to analyze “non-medical exemptions for children entering kindergarten from 2005 through 2010, and pertussis cases that were diagnosed in 2010 in California.”
The Los Angeles Times (9/30, Macvean, 3.07M) notes between 2000 and 2010, rates for “nonmedical exemptions” in California have “more than tripled, to 2.33%, with some schools reporting rates as high as 84%.” According to the researchers, clusters of exemptions and high pertussis rates “were associated with factors characteristic of high socioeconomic status such as lower population density, lower average family size, lower percentage of racial or ethnic minorities,” as well as income and other factors.
CBS News (10/1, Jaslow, 3.87M) website adds the study did not investigate the specifics of why parents refused vaccines, but did find “the families were more likely to be of high socioeconomic status.” During a press conference, director of the CDC’s National Center for Immunization and Respiratory Diseases, Dr. Anne Schuchat, said, “Clusters of people with like-minded beliefs leading them to forgo vaccines can leave them susceptible to outbreaks when measles is imported from elsewhere.”
The NPR (10/1, Shute, 465K) “Shots” blog reports that the California outbreak was not only fueled by vaccine refusal; since “pertussis is a cyclical disease,” protection seems to fade sooner than doctors originally believed. “So many older children vaccinated as youngsters were no longer immune to the bacterium. And many adults had never gotten a booster.”
Also reporting on the story are the Time (10/1, Sifferlin, 13.4M) “Healthland” blog, HealthDay (10/1, Gordon, 5K),MedPage Today (10/1, Smith, 122K), Medscape (10/1, Lewis, 164K), and the Boston.com (10/1, 1.75M) “MD Mama” blog.
Help your patients obtain affordable health insurance.
“Artificial pancreas” receives FDA approval.
The Wall Street Journal (9/27, Tadena, Subscription Publication, 5.91M) reported the Food and Drug Administration approved Medtronic Inc.’s insulin delivery pump, the MiniMed 530G “artificial pancreas.” The device is approved for use in people over 16 years of age with diabetes.
Bloomberg News (9/27, Cortez, 1.91M) noted the device’s “approval came about six months earlier than investors expected.” According to Bloomberg News, “the next generation device will be designed to anticipate blood sugar levels and administer insulin accordingly to keep patients in an ideal range with little patient involvement.”
Also reporting on the story are Reuters (9/27, Dey), the Minneapolis-St. Paul (MN) Business Journal (9/30, Grayson, Subscription Publication, 13K) “TechFlash” blog, the Minneapolis Star Tribune (9/28, Moore, 1.14M), theLos Angeles Business Journal (9/27, 98K), and Medscape (9/30, Tucker, 164K).